Apogenix’ TNFSF Receptor Agonists (HERA-Ligands)
Apogenix is developing a novel class of TNFSF receptor agonists (HERA-ligands) for the treatment of various types of cancer. Their unique molecular structure overcomes the known limitations of antibodies and other biologics targeting TNFSF receptors. Preclinical work at Apogenix has clearly demonstrated the producibility and biological activity of these novel HERA-ligands.
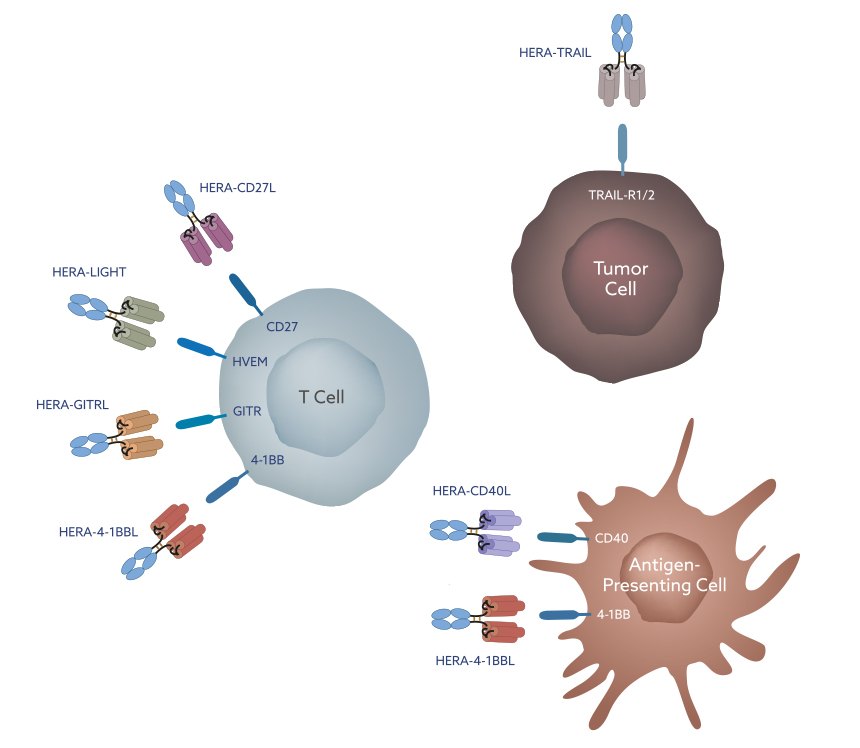
HERA-TRAIL
TRAIL receptors 1 and 2 are expressed by many different cells. The clustering of these TRAIL receptors following binding of their natural ligand leads to cell death, predominantly in tumor cells. Following the death of the tumor cell, tumor antigens are released and transported to a nearby lymph node. In the lymph node, these tumor antigens stimulate an anti-tumor immune response, potentially leading to the systemic elimination of the tumor.
The excellent antitumor efficacy of Apogenix’ TRAIL receptor agonists such as APG880 (ABBV-621) has been demonstrated in a large number of preclinical studies. Apogenix has entered into a licensing agreement with AbbVie. In Q4 2021, AbbVie initiated a clinical phase Ib study with ABBV-621 (eftozanermin-alpha) in patients suffering from multiple myeloma.
HERA-CD40L
CD40 is expressed on the surface of antigen presenting cells (APCs) as well as some tumor cell types. Binding of CD40 to its natural ligand leads to increased APC activation and an enhanced immune response.
CD40 agonist therapy plays an important role in APC maturation and their migration from the tumor to the lymph nodes, resulting in elevated antigen presentation and T cell activation.
HERA-GITRL
GITR is primarily found on the surface of T cells. It is constitutively expressed at low levels on naïve T cells and at high levels on regulatory T cells (Treg cells). Its expression is up-regulated following T cell activation. Binding of GITR to its natural ligand directly leads to increased anti-tumor T cell activation as well as their survival.
It also reduces the suppressive abilities of Treg cells, further increasing the anti-tumor immune response. These diverse mechanisms show that GITR plays an important role in initiating the immune response in the lymph nodes and in maintaining the immune response in the tumor tissue.
